http://www.meassociation.org.uk/2015/02/the-lancet-and-their-take-on-the-name-change-report-controversy-20-february-2015/
‘The Lancet’ and their take on the ‘Name Change Report’ controversy | 20 February 2015
On Feb 10, 2015, the US Institute of Medicine (IOM) published a landmark report, renaming chronic fatigue syndrome and myalgic encephalomyelitis (CFS/ME) as systemic exertion intolerance disease (SEID), and redefining the diagnostic criteria. Between 800 000 and 2·5 million Americans and an estimated 250 000 Britons have been diagnosed with CFS/ME, and the report says that the CFS/ME community and patient groups remain stigmatised.

The brief of the report was threefold: to develop evidence-based new diagnostic criteria for CFS/ME; to recommend a new name for the disorder; and to develop an outreach strategy to disseminate these changes to clinicians and the general public. The authors of the report hope that the new name will send a signal to clinicians and patients, and could be the first step towards a widespread change of attitude, through improved acceptance and understanding of the patient experience. The name itself has been constructed by the IOM to reflect the literature. Systemic implies that the condition affects many body systems; exertion intolerance is meant to convey the central feature of the disorder, although most patients are fatigued even at rest; disease implies a pathological mechanism underlying the condition, but, as the authors suggest, no disease process has yet been properly identified.
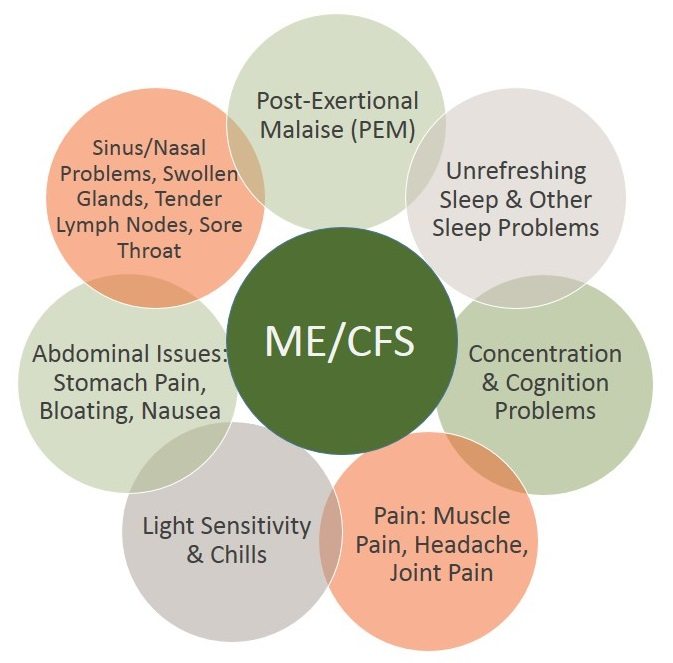
Based on a comprehensive review of data from both the research community and patient groups, the report also proposes new clinical diagnostic criteria for the disease, based around the most commonly experienced symptoms. Three core symptoms that are present for diagnosis are: impaired day-to-day functioning because of fatigue; malaise after exertion (physical, cognitive, or emotional); and unrefreshing sleep. These broad characteristics must also be accompanied by either cognitive impairment or orthostatic intolerance, or both. The authors provide good evidence for the importance of these symptoms in CFS/ME, and this clinical diagnosis might help clinicians. Doctors will still need to exclude alternative diagnoses, such as a primary sleep disorder and depressive illness; the report suggests that the presence of these diagnoses might sometimes be viewed as comorbid conditions. Some critics will say it is unfortunate that the committee has dropped a previous recommendation of undertaking a mental state examination. Additionally, the presence of orthostatic intolerance—worsening symptoms during an upright posture—should necessitate careful examination for other explanations of a patient’s symptoms. The IOM is sensible to recommend empirical testing of the new criteria, and to suggest that both the new diagnostic criteria and the name SEID will need re-evaluation within 5 years.
The causes and mechanisms behind CFS/ME have yet to be understood. The IOM was heavily and negatively lobbied by the CFS/ME community for undertaking this review. On the same day as the IOM report was released, a new Cochrane review of exercise therapy for CFS/ME was published. The Cochrane review includes results of eight randomised controlled trials, with exercise therapy ranging from 12 to 26 weeks. The conclusions of the review state that, “Patients with CFS may generally benefit and feel less fatigued following exercise therapy, and no evidence suggests that exercise therapy may worsen outcomes”. The PACE trial by Peter White and colleagues, published in The Lancet in March, 2011, was subject to widespread and unjust criticism from parts of the CFS/ME community and some advocacy groups in 2011. The PACE trial associated both cognitive behaviour therapy and graded exercise therapy with improvements in self-rated fatigue and physical function after 1 year. The most frequent criticism centred around trial outcomes and definitions, and the authors’ reply published in our correspondence columns, addressed these issues clearly. Despite critical reception at the time of publication, the results of the PACE trial paved the way for this IOM report.
The key question is whether or not these new clinical diagnostic criteria and name improve attitudes towards patients and their assessment and care. The messsage of the IOM report is that CFS/ME is a serious and complex disorder, and the authors hope that the new name could be an important step in changing perception. Further research to test these new concepts with cooperation between professional and patient groups is now needed to improve the evidence base. The IOM’s role in improving the understanding of CFS/ME, and ultimately in changing attitudes, is vital, and it is a crucial function of the IOM to constantly review and re-review the latest scientific evidence. The IOM findings and conclusions are a brave attempt to use the best available research to update the public and policy makers about a condition that causes deep community anxiety.
For the Beyond Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: Redefining an Illness report see https://www.iom.edu/Reports/2015/ME-CFS.aspx
For the Cochrane review: exercise therapy for chronic fatigue syndrome seehttp://onlinelibrary.wiley.com/enhanced/doi/10.1002/14651858.CD003200.pub3
For the PACE trial seehttp://www.thelancet.com/journals/lancet/article/PIIS0140-6736(11)60096-2/abstract
No comments:
Post a Comment